There are 90 or more plant nutrients, 17 of which are essential to plant health. Nickle was the newest to the list in the 1980’s. Without these nutrients plants cannot complete their life cycle. For a review on the role each of the nutrients plays in plant health visit 17 Essential Soil Nutrients For Plant Health. Silicon, cobalt and vanadium are also important nutrients to many plants and may eventually, with more research, be added to the list of essential plant nutrients.
Plant nutrients are grouped according to how much of that nutrient plants require. Macronutrients are nutrients that are consumed in larger quantities. As such these nutrients can easily become deficient in soils. Learning to recognize symptoms of these deficiencies and how to correct them is an important step in plant health. Micronutrients on the other hand are nutrients that are consumed in very small amounts by plants and most soils have enough of these nutrients.

Symptoms of Plant Nutrient Deficiencies
Macronutrients
Oxygen (O)
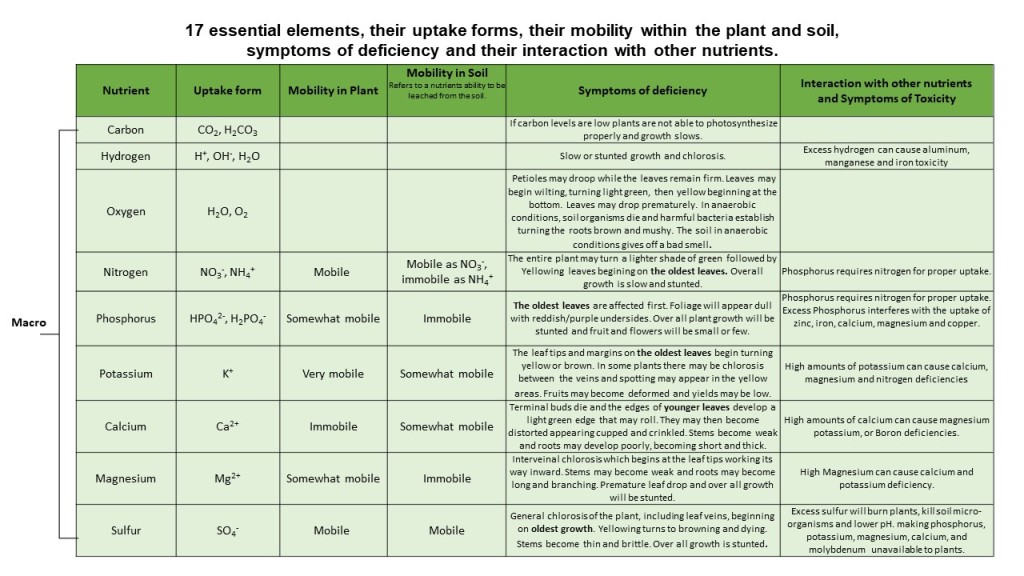
Oxygen is the second most abundant element on earth. It is essential for the processes of photosynthesis and respiration and for the storage and transference of energy within the cells of the plant. Therefore, a shortage of this nutrient causes plants to wither and die. Specific symptoms may include petioles drooping while the leaves remain firm. Leaves may begin wilting, turning light green, then yellow (chlorosis) from the base of the plant to the top beginning on the inside working out towards the outer leaves. Leaves may drop prematurely. In anaerobic conditions, soil organisms (that promote healthy soil and plants) die, and harmful bacteria establish, turning the roots brown and mushy (root rot). The soil in anaerobic conditions gives off a bad odour.
Correcting an Oxygen Deficiency
Plant roots are only able to acquire oxygen from the soil, thus, over watering, be it from rain or irrigation, is the primary reason for oxygen deficiency in plants. The water in the soil displaces the oxygen. To correct the problem, reduce watering, allow plants to dry out between watering and only give them as much water as they can drink up in a couple of hours. Break up any soil compaction and hard pan and work plenty of organic matter into the soil to improve drainage. Check the soil grade and make any corrections. Regularly monitor your soil moisture, especially after a period of heavy rains. Once roots begin to rot the damage is usually irreversible.
Hydrogen peroxide (H2O2) can give plants an added boost of oxygen and protect them against root rot. Using a 3% solution of hydrogen peroxide, add 2-3 tsp. to 1 liter of water or 2 tbsp. per gal. of water and use as a soil drench.
Carbon (C)
Carbon is the fourth most abundant element in the Universe and deficiencies are rare in an outdoor setting. Like oxygen, carbon is essential in the process of photosynthesis, whereby the plant, in the presence of sunlight, takes in carbon dioxide and water and creates oxygen and energy in the form of sugar. Thus, if carbon levels are low plants are not able to photosynthesize properly and growth slows.
Correcting a Carbon Deficiency
In a grow room setting you may occasionally experience carbon deficiency. To correct this, install a fan with ventilation to the outside to bring in a fresh supply of carbon dioxide. Even having people and animals around your plants helps to provide them with the carbon dioxide that they exhale. For larger operations there are carbon dioxide generators.
In an outdoor setting there is no shortage of carbon in the atmosphere, but to move and hold that carbon into the soil (where it does not contribute to global warming), anything you can do to improve soil health will help to increase carbon in the soil. Checkout my article on Healthy Soil for tips and information on creating a healthy soil.
Carbon Toxicity Symptoms:
Carbon toxicity is extremely rare. Typically, the more carbon, the more growth. However, it does place other demands on the plants, like increased water and nitrogen needs. In those rare cases of carbon toxicity plants leaves develop zebra like stripes on them and yields are reduced.
Hydrogen (H)
“Hydrogen is the most widely distributed element in the world, accounting for more than 75% of the mass of the universe” (Zeng, J., Ye, Z. & Sun, X (2014)). It is the smallest and lightest of the elements and in its atomic form is highly unstable and combustible. The majority of hydrogen however exists in its molecular form H2, a stable molecule. Hydrogen (H2) is strongly attracted to oxygen and readily attaches to it, forming water (H2O). This is how plants predominantly acquire hydrogen.
Hydrogen is the 3rd key player in the process of photosynthesis and respiration and is required for the manufacturing of food energy for the plant and thus it’s growth. Plants deficient in hydrogen will exhibit slow or stunted growth. The role of hydrogen in plants has also been linked to stress resistance (such as salinity, drought, cold and heavy metals), seed germination, regulation of flowering time, improved crop resistance to disease and pests, reduced fertilizer use, and crop preservation (Zeng, J., Ye, Z. & Sun, X (2014)). Plants deficient in hydrogen may be experiencing drought and may exhibit drought like symptoms such as wilting, yellowing of leaves, brown leaf margins or premature leaf drop, branch dieback, damaged roots, even death. Without enough water plants will also be unable to take up soil nutrients and may exhibit symptoms of those deficiencies as well.
Correcting a Hydrogen Deficiency
Water your plants sufficiently. Add organic matter to the soil, which is naturally high in hydrogen, and which will also help to improve the storage and movement of water. Examine the roots of suspected plants to ensure they are not damaged, diseased, pot bound or girdled, as plants take up water through their roots. A product called hydrogen water, which is regular water with dissolved hydrogen gas (H2) added to it, can also be watered into the soil.
Hydrogens Role in Soil pH:
Hydrogen ions play a role in a soil’s pH level, which in turn affects the availability of soil nutrients, making some nutrients more readily available and others less available. The element H and the molecule H2 hold a neutral charge and as such do not affect the pH of soil or water; but as hydrogen adds or drops electrons, the hydrogen ions can become positively or negatively charged. A negatively charged hydrogen ion has an acidifying effect while a positively charged hydrogen ion has an alkalizing effect. Most nutrients are available in the 6.0 to 7.0 range. Therefore, maintaining a healthy pH range is critical to plant health and growth. Altering a soil’s pH level can be a slow process affected by soil texture, temperature, moisture levels and the organic content in the soil. To lower soil pH add elemental sulfur, sulfuric acid, ferrous sulfate or aluminum sulfate. Elemental sulfur is safe but slow to lower pH, requiring 6 months or more for soil bacteria to convert it to sulfuric acid. Ferrous sulfate (which also will add iron to the soil) and aluminum sulfate (which can be toxic to some plants) are quicker. Sulfuric acid applications are the fastest, but dangerous and require specialized equipment and specially trained applicators. To raise a soils pH add ground agricultural limestone or other form of lime.
Before you begin adding fertilizers to the soil make sure other factors such as cool soil temperatures, nutrient imbalances, soil compaction, soil water content (to wet or to dry) and soil pH are not the cause of the deficiency. Correct these problems first, then test your soil for the suspected deficient nutrient(s) and adjust accordingly.
Nitrogen
Nitrogen is a mobile nutrient and as such the oldest leaves are affected first when there is a deficiency. This is due to the younger leaves being able to move those nutrients up from older leaves when there is a shortage. Eventually however if the deficiency persists the new leaves will eventually become affected as well. Nitrogen is essential for photosynthesis and promotes a deep green colour and lush growth. Thus, a visual symptom of a nitrogen deficiency is leaves becoming lighter green in colour, then lower leaves that begin to yellow, sometimes down the middle first, then eventually the whole leaf yellows followed by browning then dying. Sometimes there is some reddish discolouration as well. Overall plant growth will be slow or stunted.
Correcting a Nitrogen Deficiency
Although the earth’s atmosphere is composed of 78% nitrogen (N2), it does not exist in a form that plants can take up nitrate (NO3–) or ammonium (NH4+). Plants require a great deal of nitrogen in order to grow and as such this nutrient is often deficient in soils. Nitrogen can be added to the soil in one of 3 ways: lightening, artificially manufactured nitrogen fertilizer (of which there are many types, each with its benefits and drawbacks) and biological fixation: which occurs when organic materials, that are in the soil, are decomposed by various soil micro-organisms and bacteria making available nitrogen in the form plants can take up. Examples of organic materials are animal manures, composts, blood meal, fish emulsion, plant material, fish meal, cottonseed meal, soybean meal, alfalfa meal and green manure crops. Another type of fixation occurs in legumes when rhizobia bacteria take atmospheric nitrogen (N2) and break it down to a form that plants can take up (NO3–) then store that nitrogen on the plant’s roots in nodules.
Many factors affect the uptake of nitrogen including the stage of development of the plant(s), temperature, moisture, soil pH, soil texture, soil structure, drainage, salt content and wind.
As a general guide:
- Apply your nitrogen source when the plants are most actively growing to reduce volatilization (the loss of nitrogen to the atmosphere as ammonia gas).
- On high pH soil that is prone to nitrogen volatilization choose a slow-release fertilizer such as organic sources or ammonia (NH3) and stay away from nitrates (that can raise your soil pH).
- For acidic soils nitrates will raise pH as will an application of lime which then helps the plants to better take up nitrogen (and other nutrients).
- Urea (CO(NH2)2) based fertilizers are prone to leaching (being washed out of the root zone) and volatilization (lost to the atmosphere as ammonia gas). To help prevent this make sure the urea is worked or well-watered into the soil. Keep the soil evenly moist. It takes a couple of weeks (more quickly on high pH soils) for the urea to convert to NH3 then NH4 and finally NO3- for plant uptake. Urea has neither a positive nor negative charge and does not have an impact on soil pH. Do not apply during hot, humid weather.
- Plants require less nitrogen at the flowering stage.
- Anhydrous ammonia will temporarily reduce populations of soil microorganisms.
Symptoms of Nitrogen Toxicity:
The leaves become unusually dark green, and the leaf tips burn and turn brown. Symptoms begin on the lower leaves.
Phosphorus
Symptoms of Phosphorus Deficiency:
As a mobile nutrient the oldest leaves are affected first. Foliage will appear dull with reddish/purple undersides. Overall plant growth will be stunted and fruit and flowers will be small or few. In severe cases there can be necrosis and leaf drop.
Correcting a Phosphorus Deficiency
Phosphorus takes a long time to break down and moves through the soil profile very slowly. As such it is rarely deficient in our soils. Sometimes what appears to be a phosphorus deficiency is actually a nitrogen deficiency, as nitrogen is needed for proper uptake of phosphorus. Applying a layer of organic matter such as compost, shredded leaves or manure will help serve to add phosphorous to soils. Additional sources include bone meal and phosphate rock, especially suitable in low pH and low calcium soils. Try to stay away from super phosphate as it contains harmful salts that can cause an imbalance with your soil’s microorganisms. There are no synthetic sources of phosphorous available.
Symptoms of phosphorus toxicity:
Excess phosphorus inhibits the uptake of micronutrients like iron, zinc and manganese and kills off beneficial mycorrhizal fungi. It also contributes to algae blooms when the excess phosphorus is washed out of the soil into ground water.
Potassium Deficiency:

The leaf tips die and the margins on the oldest leaves begin turning yellow then brown. In some plants there may be chlorosis between the veins and spotting may appear in the yellow areas. Fruits may become deformed, and yields may be low. Plants will be less heat tolerant and more prone to insect and disease problems.
Correcting a Potassium Deficiency
Excess calcium, sodium, or nitrogen can prevent plants from absorbing potassium, adjust your fertilizer program accordingly. There are several sources of natural and organic potassium available to homeowners such as: seaweed, cocoa bean hulls, manure, compost, dried banana peels, hardwood ashes, granite dust, kelp meal, and green sand. Potash is a commonly used source of potassium and is commercially available is several forms, such as potassium sulfate (Sulfate of potash), potassium nitrate and muriate of potash (potassium chloride).
Symptoms of Potassium Toxicity
Excess potassium does not appear to have a toxic effect on plants. It can induce deficiencies of other nutrients however, particularly nitrogen, calcium and magnesium.
Calcium (Ca)
Calcium is an immobile nutrient; meaning is unable to move well within the plant. As such the first symptoms of a calcium deficiency tend to occur at the shoot and root tips. Terminal buds die and the edges of younger leaves develop a light green edge that may roll. “Newly emerging leaves may stick together at the margins, which causes tearing as the leaves expand and unfurl” (Uchida R. 2000). They may then become distorted appearing cupped and crinkled. Older leaves become dull, and the leaf margins turn brown. Stems become weak and roots may develop poorly, becoming short and thick (University of Guelph (n.d.).
Other symptoms may include abnormally dark green foliage, premature shedding of blossoms and buds (or blossom end rot in tomatoes, peppers and water melons). For more information about calcium check out my article on Calcium It’s Role in Soil and Plant Health.
Correcting a Calcium Deficiency
Calcium deficiency is rare on soils formed from limestone and calcareous parent materials. It most commonly occurs on sandy or acidic soils and soils with low cation exchange capacity (CEC). If your pH is too acidic (below 6) first work at alkalizing the soil, then add some bone meal which high in calcium. On sandy soil add healthy amounts of compost to help improve the nutrient holding capacity of the soil. Natural sources of calcium include weathered soil minerals such as calcite, dolomite lime, feldspar and gypsum, as well as crushed oyster shells, eggshells, chamomile and seaweed. Applied sources include lime (Calcium carbonate), Calcium chloride (used as a foliar spray to treat blossom end rot, tip burn of cabbage, bitter pit, and cracking cherries) and gypsum (Calcium sulfate) (the latter two will not raise soil pH). Note: Lime and gypsum have low solubility and take a long time to break down in the soil (months to years), best to apply them in the fall, to be ready for spring planting.
Symptoms of Calcium toxicity:
Calcium toxicity is rare, but it can raise soil pH which can affect the uptake of other nutrients. Calcium also competes with magnesium, boron and potassium on the soil exchange and makes them less available to plants. In a soil toxic in calcium, seeds may not germinate, and plant growth will be reduced.
Magnesium (Mg)
Magnesium is a mobile nutrient and symptoms will begin at the base off the plant on the oldest leaves. The most typical symptom is interveinal chlorosis (the veins stay green while the tissue between them losses it’s green colour). Affected leaf tissue may be yellow, bronze, reddish or even white, depending on the species. There may be some necrotic leaf spots as well. Stems may become weak, and roots may become long and branching. As the deficiency continues leaf edges will begin dying and may curl upward. Whole leaves will die and drop prematurely. Overall growth will be stunted.
Correcting Magnesium Deficiency
The soils most likely to be deficient are sandy or acidic soils. Cold wet soils may also be temporarily deficient in this nutrient. Ideal soil pH for magnesium absorption is between 6.0 and 6.5. If your soil pH is off, work at correcting that first before adding fertilizer. Excessive amounts of calcium or potassium in the soil can lock up magnesium. Conversely an excess of magnesium will lock up potassium, zinc, boron and manganese. It can also interfere with soil structure making it sticky. Adjust your overall fertilizer program to accommodate. Sources of magnesium include Dolomitic limestone, Epsom salts (magnesium sulfate), K-MAG (sulfate of potash-magnesia).
Symptoms of Magnesium Toxicity:
Magnesium toxicity symptoms are quite rare. Plants are more likely to exhibit the nutrient deficiencies that excess magnesium causes.
Sulfur (S)
General chlorosis of the plant, including leaf veins, beginning on oldest growth. Yellowing turns to browning and dying. Stems become thin and brittle. Overall growth is stunted.
Correcting Sulfur Deficiency
Sulfur deficiency is most likely to occur in cold, sandy soils that are low in organic matter and receive a fair amount of rainfall. Generally, though, sulfur deficiency is not common. Begin by incorporating healthy amounts of organic matter into the soil, which will help to improve the nutrient holding capacity of the soil and is of itself a source of sulfur. Other sources of sulfur include gypsum, Epsom salts, elemental sulfur, iron sulfate and rainwater.
Symptoms of Sulfur Toxicity:
Excess sulfur will burn plants, kill soil micro-organisms and insects. High sulfur soils have a low pH making other nutrients on the soil exchange (like phosphorus, potassium, magnesium, calcium, and molybdenum) unavailable to plants.
MICRONUTIENTS
Sources of Micronutrients: Organic matter is an excellent source of micronutrients. Spreading a layer of compost or manure every spring will usually provide adequate amounts of all of these nutrients. Deficiencies are more likely to occur on sandy soils, acid soils with abundant rainfall or in very alkaline soil. Correcting soil pH is the key to unlocking these nutrients. Other sources of micronutrients are liquid seaweed, granite dust, chelates of the mineral and rock phosphate.
Boron (B)
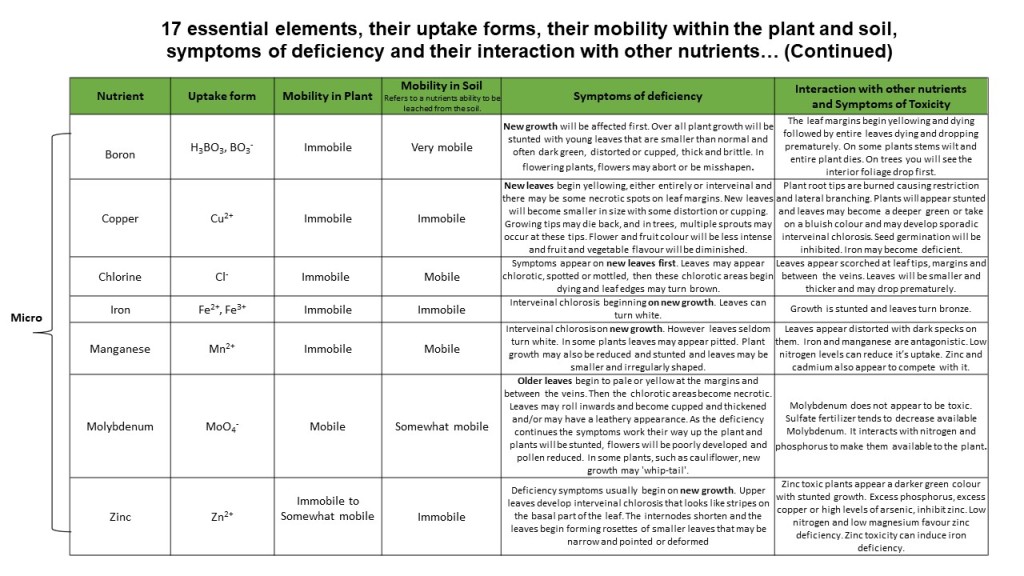
Boron is immobile within the plant; thus new growth will be affected first. Overall plant growth will be stunted with young leaves that are smaller than normal and often dark green, distorted or cupped, thick and brittle. In flowering plants, flowers may abort or be misshapen. Many boron deficiency symptoms are specific to the particular plant grown. In some species (like roses) there is loss of apical dominance resulting in multiple branching. In pines the central leader dies. If you suspect boron deficiency research the specific symptoms for your plant.
Correcting Boron Deficiency
If you suspect a boron deficiency have a soil test done to confirm and guide you in correcting it. Boron is a tricky nutrient with a narrow acceptable range; not enough and plants suffer, to much and you can kill them. Boron is typically present enough in soils but of the 8 micronutrients it is the one most likely to be deficient. Naturally occurring boron is in the form of borates, which are inorganic salts that contain boron. They are widely found in nature, and are present in oceans, sedimentary rocks, coal, shale and some soils. Applied sources include: borax (such as 20 Mule Team Borax applied at a rate of 1 tbsp. per 100 linear feet), boric acid (which can be found in drinking water at different levels) and Solubor (which is used as a foliar spray on high pH soils).
If your drinking water or soil contain high levels of boron try growing plants that are more boron tolerant like roses, lavender, yarrow, grapevines, red hot poker, broccoli, cauliflower, beets, turnips, cabbage, brussels sprouts, parsley, apples, pears and grapes. These are also the same plants you are likely to see a deficiency on in low boron soils.
Symptoms of Boron Toxicity:
The leaf margins begin yellowing and dying followed by entire leaves dying and dropping prematurely. On some plants stems wilt and entire plant dies. On trees you will see the interior foliage drop first. Symptoms can vary depending on the plant.
Copper (Cu)
Copper is immobile within the plant and as such the new leaves are affected first. Leaves yellow, either entirely or interveinal and there may be some necrotic spots on leaf margins. New leaves will become smaller in size with some distortion or cupping. The growing tips may die back, and in trees, multiple sprouts may occur at these tips, giving it a bushy appearance. Flower and fruit colour will be less intense and fruit and vegetable flavour will be diminished.
Correcting Copper Deficiency
Copper deficiency is not common and where it occurs it can often be attributed to excess potassium, phosphorus or other micronutrients. High pH soils can also prevent the uptake of copper. Begin by testing your soil for nutrient imbalances and pH and correct those before adding copper to the soil. Copper deficiency is most likely to occur on Peaty, or sandy soils and soils with high concentrations of organic matter. Organic sources of copper include sewage sludge and animal manures (especially pig manure). Sources of applied copper include Copper sulfate, copper oxide, and copper chelate. Copper persists in the soil for years due to it’s low solubility so take care when using fertilizers and fungicides (like bordeaux) containing copper.
Symptoms of Copper Toxicity:
Copper toxicity primarily affects the plant roots restricting their growth by burning the root tips, causing lateral branching. Plants will appear stunted, and leaves may become a deeper green or take on a bluish colour and may develop sporadic interveinal chlorosis with the chlorotic areas becoming pale and slightly sunken. Seed germination will be inhibited. Iron may become deficient, and plants may exhibit symptoms of that and other nutrient deficiencies.
Chlorine (Cl)
Chlorine (Cl) is an extremely unstable chemical element. In order to stabilize itself, it forms a bond with another chlorine (Cl) atom to form a Chlorine gas (Cl2). This diatomic molecule (Cl2) then converts to chloride (Cl-) in the soil, a stable molecule, in the form plants can take up.
Symptoms of Chlorine Deficiency
As a mobile nutrient, within the plant, symptoms typically appear on older leaves first. Leaves may appear chlorotic, spotted or mottled, then these chlorotic areas begin dying and leaf edges may turn brown. There typically is abrupt boundaries between the affected and healthy tissue (Wade K. M, 2019. In some plants (such as tomato) leaf edges roll upward. Plants may wilt (leaves wilt at the margins) and roots become restricted and highly branched.
Correcting Chloride Deficiency
Chloride deficiency is not common especially in coastal areas but may become deficient in sandy soils further inland, especially if they receive a good deal of water. Chloride is not held on the soil exchange and as such is easily leached from the soil, especially sandy soils. To help hold it in the soil add plenty of organic matter. Care must be taken not to add to much chloride to the soil as it can be toxic to plants and cause potassium and phosphorus deficiency. Several trees are sensitive to chloride such as ash, boxelder, Siberian crabapple, dogwood, horse-chestnut, silver maple, sugar maple, pin oak, sweetgum, and yellowwood. There are also several fruit and veg crops that are sensitive to chloride such as beans, citrus, potatoes, lettuce, strawberry, pepper, avocado and almonds. Foliar sprays used for deficient plants advisable when working around other sensitive plants.
Sources of natural chloride include sea spray, rainwater, irrigation water, organic matter, underground rock formations, dust and air pollution. Sources of applied chloride include: Potassium chloride (KCl), known as muriate of Potash (MOP); Calcium chloride (de-icing salt), which also serves to raise soil pH. ; Magnesium chloride (Epsom salt) which is a harvested product found in well water and permitted in organic farming (sold under trade names like AgMag, NutriMag); Ammonium chloride (a nitrogen fertilizer with chloride) and Sodium chloride (including table salt, sea salt, himalayan salt and rock salt), not certified organic.
Symptoms of Chloride Toxicity:
Although chloride is easily leached away, coastal gardens and gardens near chlorinated pools, may become toxic. Leaves that appear scorched or burned (or in some cases bleached) is a classic symptom. The scorched areas appear at leaf tips, margins and between the veins. Leaves will be smaller and thicker and may drop prematurely. Overall plant growth will decline.
To reduce chloride in the soil, flush the area well with water and work some gypsum into the soil at a rate of about 50 lbs. per 1000 square feet, in loam soils. Less gypsum is needed in sandy soils, more in heavy clay soils.
Iron (Fe)
Iron is not a mobile nutrient within the plant and as such symptoms appear first on newer leaves. Interveinal chlorosis is the most notable symptom. The leaves may become so chlorotic between the veins that they turn white. Additional symptoms that are less common include stunted or malformed new growth and leaf, bud or fruit drop. Foliage may also develop brown leaf margins.
Correcting Iron Deficiency
Iron is the 4th most abundant element on earth (Schulte E.E. n.d.). As such, it is usually abundant in most soils. Deficiencies usually are the result of iron being locked up in the soil and not accessible to plants. The factors that most affect irons availability are soil pH, soil aeration and organic matter. In the soil, iron exists in the ferrous Fe++ or ferric Fe+++ forms. The ferric forms are extremely insoluble. In high pH soil (alkaline) the iron forms ferric compounds while in low pH soil (acidic) iron forms ferrous compounds, that are more available to plants. Thus, lowering your soil pH, if it is high, is helpful to unlocking iron and correcting an iron deficiency.
Ensuring your soil is well aerated by avoiding over watering, correcting drainage problems and breaking up compaction also helps to make iron more available to your plants. Adding organic matter to the soil also helps to both provide a source of iron and to make existing soil iron more available to plants.
There are several sources of applied iron, and you may have to do a little homework to decide which one is right for you. As a general guide though iron chelates such as Fe-DTPA, Fe-EDDHA, EDDHSA, Fe-EDTA, Fe-EDDHMA and Fe-HEDTA, prevent the oxidation of iron, making it more available to plants. They are available in several forms such as pellets, spikes, powders and granular. Often, they are applied as a foliar spray, which provides a fairly fast green up of the leaves, but may not last, and require several applications. I have highlighted Fe-EDDHA as research has found that one to be least effective due to it not remaining stable in the soil beyond a 6.3 pH (Schulte E.E. n.d.). Other elements present in the soil (such as calcium) can also impact the uptake of iron chelates.
Symptoms of Iron Toxicity
Excess available iron in a soil will stunt growth and cause leaves to turn bronze. To correct iron toxicity lime acidic soils to increase their pH, avoid adding organic matter or any additional sources of iron.
Some plants that are prone to iron toxicity include: zonal geraniums, lisianthus, African marigolds, pentas, New Guinea impatiens and lilies (Brubaker V. 2016).
Manganese (Mn)
Manganese deficiency looks very much like iron deficiency with interveinal chlorosis on new growth. However, leaves seldom turn white with manganese deficiency. In some plants leaves may appear speckled or pitted (specks with depressed areas around them). Plant growth may also be reduced and stunted, and leaves may be smaller and irregularly shaped.
Correcting Manganese Deficiency
Manganese deficiency is common on soils with a pH above 6.5 and on soils high in organic matter. Waterlogged soils will also be deficient. Foliar applications of either manganese sulfate or chelated manganese appear to be the most effective at relieving a manganese deficiency. Several applications may be required.
Interactions in the soil with other nutrients (calcium, magnesium, and Iron) can also induce a manganese deficiency. Iron and manganese are antagonistic and thus excess available iron will reduce available manganese. Low nitrogen levels can reduce manganese uptake by plants. Applying a general fertilizer (which is usually low in micro-nutrients) will help to improve the uptake of manganese and increase the concentration of anions (negatively charged molecules) in the soil. Zinc and cadmium also appear to compete with manganese (Adamczyk-Szabela, et al., 2020). Manganese also serves to increase phosphorus and calcium availability.
Symptoms of Manganese Toxicity
Leaves appear distorted with dark specks on them. In severe cases there is necrosis beginning on the leaf edges and working in.
Molybdenum (Mo)
Molybdenum is essential for nitrogen uptake and the fixation of atmospheric nitrogen in legumes. As such symptoms of molybdenum deficiency appear very similar to nitrogen deficiency. Older leaves begin to pale or yellow at the margins and between the veins. Then the chlorotic areas become necrotic. Leaves may roll inwards and become cupped and thickened and/or may have a leathery appearance. As the deficiency continues the symptoms work their way up the plant and plants will be stunted, flowers will be poorly developed, and pollen reduced. In some plants, such as cauliflower, new growth may ‘whip-tail’.
Correcting a Molybdenum Deficiency
Molybdenum is a silver-grey metal that is released from solid minerals through normal weathering processes and converts to molybdate (MoO42- ) in the soil (the form plants can take up). Plants only require a very small amount of molybdenum and thus most soils have enough in them. The soils most likely to be deficient are sandy soils that received a lot of precipitation and soils with a low pH. If you have a sandy soil, add plenty of organic matter to help hold the nutrient in and which is of it’s self a source of molybdenum. On acidic soils work at increasing soil pH to make molybdenum more available to the plant. Another factor to consider is the plants source of nitrogen. Molybdenum converts nitrate to ammonia within the plant. Thus, if your nitrogen source is ammonia less molybdenum will be required and conversely if your nitrogen source is nitrate, more molybdenum will be required by the plant to convert it.
Natural sources of molybdenum include organic matter and seaweed extract. Applied sources include: Ammonium Molybdate (H8MoN2O4) and Sodium Molybdate. (Na2MoO4). They are highly soluble in water and are often used as foliar sprays, although they can also be used in the soil and for seed treatment. Molybdenum trioxide (MoO3) has low solubility and is used only as a slow release soil fertilizer.
Molybdenum deficiency is most commonly found on nitrogen fixing plants and on the brassica family.
Symptoms of Molybdenum Toxicity
Molybdenum does not appear to be overly toxic to plants.
Zinc (Zn)
Deficiency symptoms usually begin on new growth. Upper leaves develop interveinal chlorosis that looks like stripes on the basal part of the leaf. The internodes shorten and the leaves begin forming rosettes of smaller leaves that may be narrow and pointed or deformed. In flowering plants flower buds may die off. In fruit trees, yields will be reduced, and fruit may be deformed.
Correcting a Zinc Deficiency
Zinc deficiency is more common than the other micronutrients and several factors can contribute to its deficiency such as: high soil pH, low organic matter, sandy soils, over watering, excess phosphorus, excess copper, low nitrogen, low magnesium, high levels of arsenic and cold wet soil conditions. New subdivisions that have been leveled and had their topsoil removed may be deficient, especially if the soil has also been compacted by the heavy equipment. Begin correcting your zinc deficiency by first addressing and correcting the above conditions, especially adding in plenty of organic matter and lowering a high soil pH. Have a soil test done so you can determine the extent of the deficiency and which nutrients are out of balance.
Natural sources of zinc include tap water, organic matter and kelp extract. Applied sources include Zinc Chelates (which are organic and may be applied as foliar sprays or to the soil), micro-nutrient foliar sprays that contains zinc, Zinc Sulfates (hydrated), Zinc Oxy-sulfate and Zinc-Ammonia Complexes. Zinc can be toxic so be sure to have a soil test done first before adding zinc fertilizer to the soil.
Symptoms of Zinc Toxicity
Zinc toxicity is more likely to occur on an acidic soil. Factors that contribute to excess zinc are high magnesium levels and pollutants with high metal content such as sewage sludge and deteriorating metal equipment. Symptoms of zinc toxicity are subtle and hard to diagnose. Leaf colour will be a darker green and growth will be stunted. In some plants there may be red pigmentation, particularly on petioles, down the veins and around leaf margins. Iron deficiency may also be induced.
Nickle (Ni)
As mobile nutrient symptoms appear first in old growth. One of the most common symptoms is leaflet-tip necrosis (leaf tips dying back). Symptoms can vary depending on plant species. “In woody ornamentals, symptoms occur in the spring in new emerging growth and may include shortened internodes (giving a rosetting appearance to the plant), weak shoot growth, death of terminal buds and eventual death of shoots and branches” (Buechel T. 2021). In some tree species (such as pecan) the leaves become “mouse eared”. In nitrogen fixing plants such as beans, peas, lupins, false indigo, sweet peas, white and red clover, alder, golden chain tree, black locust, redbud, sea buckthorn, mimosa and so many more from the fabaceae family there is delayed nodulation on roots and reduced efficiency of nitrogen fixation causing symptoms such as whole leaf chlorosis along with leaf tip necrosis.
Correcting a Nickel Deficiency
Nickel deficiency is most likely to occur on high pH soils, where it tends to oxidize to unavailable forms. If you have a high pH work at lowering that to near neutral, or ideally for nickel, 6.7 or less. Elemental sulfur can be useful for this purpose. Competing plant nutrients include: Calcium, Magnesium, Zinc, Copper, Iron and Manganese. Have a soil test done to determine any nutrient imbalances, then work at correcting those.
Natural sources of nickel include sewage sludge, some manures (especially horse). Sources of applied nickel include nickel sulfate or chelated nickel (usually as foliar sprays). Nickel can also be found as a contaminant in fertilizer and the irrigation water. Great care should be taken when working with nickel fertilizers as they can become toxic to plants even in small amounts and can contaminate irrigation water. “Nickel sulfate is also known to cause severe dermatitis, skin and asthma-like allergies and affects the lungs, kidneys, gastrointestinal tract and neurological system. Nickel sulfate is a known carcinogen and is associated with an increased risk of developing lung and nasal cancers (Hassan MU et al. 2019).
Symptoms of Nickel Toxicity
An excess of nickel can reduce seed germination, restrict root growth and cause iron, zinc or copper deficiency. New leaves will exhibit interveinal chlorosis, then the necrotic areas will begin dying out beginning at the margins working in. Eventually the entire plant will die.

Summary
Of the many plant nutrients, scientists have determined that the preceding 17, are absolutely essential for all higher plant life. Extensive research has been carried out to determine not only the specific role each nutrient plays but also what deficiency symptoms look like in plants. Learning to recognize these symptoms and how to correct them is an important step in plant health. For more information check out the links below, or any number of other credible sources available in book form or on the internet.
References
Adamczyk-Szabela D., Lisowska K., Romanowska-Duda, Z., M Wolf W.M., (2020). National Library of Medicine. https://pubmed.ncbi.nlm.nih.gov/32015369/
Blaszczak-Boxe A., (2015). Facts About Hydrogen. https://www.livescience.com/28466-hydrogen.html
Bloodnick E., (2021). Role of Manganese in Plant Culture. Pro-mix Training Center. https://www.pthorticulture.com/en/training-center/role-of-manganese-in-plant-culture/
Bloodnick E., (2021). Role of Zinc in Plant Culture. Pro-mix Training Center. https://www.pthorticulture.com/en/training-center/role-of-zinc-in-plant-culture/
Bloodnick. E, (2021). Role of Copper in Plant Culture. Pro-mix Training Center. https://www.pthorticulture.com/en/training-center/role-of-copper-in-plant-culture/
Brown P.H., Welch, R. M., & Cary, E. E. (1987). Nickel: A Micronutrient Essential for Higher Plants. Plant Physiology, 85(3), 801–803. http://www.jstor.org/stable/4270996
Brubaker V., (2016). Feast or Famine: Managing Iron Deficiency and Toxicity. Growers Talk. https://www.growertalks.com/Article/?srch=1&articleID=22052&highlight=feast+or+famine++managing+iron+deficiency+and+toxicity
Buechel T., (2021). Role of Nickel in Plant Culture. Pro Mix Training Center. https://www.pthorticulture.com/en/training-center/role-of-nickel-in-plant-culture/
Canola Council of Canada, (2016). Canola Watch, issue 3, Nitrogen review: Sources and plant availability. Nitrogen review: Sources and plant availability
Earth Crew Inc., (n.d.) Nitrogen Organic Plant Fertilizer. https://earthcrew.com/nitrogen-organic-plant-fertilizer/
Fite K., (n.d.). (PhD, Plant & Environmental Science). Boron Toxicity. Bartlett Tree Experts. https://www.bartlett.com/resources/technical-reports/boron-toxicity
Guodong Liu, E. H. Simonne, and Yuncong Li. (2020). (Horticultural Sciences Department). NICKEL NUTRITION IN PLANTS. U.F/ask ifas. Publication #HS1191. http://www.nutricaodeplantas.agr.br/site/downloads/unesp_jaboticabal/Niqueldeficiencia1.pdf
Dr. Hadrami A. 2018. OMEX. KNOW YOUR NUTRIENTS: 16 ESSENTIAL ELEMENTS. https://omexcanada.com/plant-nutrition/know-your-nutrients/16-essential-elements
Hassan MU, Chattha MU, Khan I, Chattha MB, Aamer M, Nawaz M, Ali A, Khan MAU, Khan TA., (2019). Nickel toxicity in plants: reasons, toxic effects, tolerance mechanisms, and remediation possibilities-a review. Environ Sci Pollut Res Int. 2019 May;26(13):12673-12688. doi: 10.1007/s11356-019-04892-x. Epub 2019 Mar 29. PMID: 30924044. https://pubmed.ncbi.nlm.nih.gov/30924044/
Hyo technologies, (n.d). Question and Answer. https://hyotechnologies.com/q—a.html
Liu G., Simonne E. H., and Li Y., (2020). (U of F, Horticultural Sciences Department). NICKEL NUTRITION IN PLANTS. https://edis.ifas.ufl.edu/publication/hs1191
Madhusha, (2017). Relationship Between Hydrogen Ions and pH. Pediaa. https://pediaa.com/relationship-between-hydrogen-ions-and-ph/
Mattson N., (Assistant Professor and Floriculture Extension Specialist, Cornell), Leatherwood R. , (Postdoctoral Associate, Cornell) and Peters C. (J.R. Peters Inc). Nitrogen: All Forms Are Not Equal. Cornell University Cooperative Extension Reprinted from the June 2009 Edition of GMPro Magazine. http://greenhouse.cornell.edu/crops/factsheets/nitrogen_form.pdf
Mosiac, (n.d.) Nutrient Knowledge, Manganese. https://www.cropnutrition.com/nutrient-knowledge
Molecular Hydrogen Institute. (n.d.). Dummies Guide To Hydrogen. http://www.molecularhydrogeninstitute.com/dummies-guide-to-hydrogen
Mosiac, (n.d.) MANGANESE IN CROP PRODUCTION., https://www.cropnutrition.com/resource-library/manganese-in-crop-production
Paungfoo-Lonhienne C., Thierry G. A. Lonhienne T.G.A., Rentsch D., Robinson N., Christie M., Webb, R.I., Gamage H.K., Carroll B.J., Schenk P.M., Schmidt S., (2008). Plants can use protein as a nitrogen source without assistance from other organisms, Proceedings of the National Academy of Sciences Mar 2008, 105 (11) 4524-4529; DOI: 10.1073/pnas.0712078105. https://www.pnas.org/doi/10.1073/pnas.0712078105
Princeton University, (2019). “Nature’s backup plan for converting nitrogen into plant nutrients.” ScienceDaily. http://www.sciencedaily.com/releases/2019/11/191111180100.htm
Queensland Gov. (updated September 2013). Soil pH. https://www.qld.gov.au/environment/land/management/soil/soil-properties/ph-levels
Rens L., PhD, (n.d.) 10 Things to Know About Chloride, Intrepid Potash. https://www.intrepidpotash.com/2020/05/01/10-things-to-know-about-chloride/
Riebeek H., (2011). The Carbon Cycle. Nasa Earth Observatory. https://earthobservatory.nasa.gov/features/CarbonCycle/page1.php
Rosales M, Franco-Navarro J, Peinado-Torrubia P, Díaz-Rueda P, Álvarez R, Colmenero-Flores J., 2020. PubMed, Chloride Improves Nitrate Utilization and NUE in Plants. https://pubmed.ncbi.nlm.nih.gov/32528483/
Schulte E.E., n.d., Soil and Applied Iron. Understanding Plant Nutrients. A3554. http://corn.agronomy.wisc.edu/Management/pdfs/a3554.pdf
Uchida R., (2000). Essential Nutrients for Plant Growth: Nutrient Functions and Deficiency Symptoms. Chapter 3. From: Plant Nutrient Management in Hawaii’s Soils, Approaches for Tropical and Subtropical Agriculture. J. A. Silva and R. Uchida, eds. College of Tropical Agriculture and Human Resources, University of Hawaii at Manoa, ©2000. https://www.ctahr.hawaii.edu/oc/freepubs/pdf/pnm3.pdf
University of Guelph (n.d.). Introduction to Soil Science and Plant Growth Resources. Appendix C. Macronutrients and Micronutrients Source, Functions and Deficiency/Toxicity Symptoms
USDA, (n.d.). Soil Nitrogen. Soil Quality Kit- Guide for Educators. United States Department of Agriculture, Natural Resources Conservation Services. https://www.nrcs.usda.gov/Internet/FSE_DOCUMENTS/nrcs142p2_053274.pdf
Water Matters, (2015). Hydrogen: What’s the difference between H, H2, H+, H- and OH- ? Water Treatment Guide. https://yourwatermatters.com/hydrogen-whats-the-difference-between-h-h2-h-h-and-oh/
Woodstream Corporation, Inc., (n.d.) Safer’s Brand, Cures For Plant Nutrient Deficiencies. https://www.saferbrand.com/articles/cures-plant-nutrient-deficiencies
Zeng, J., Ye, Z. & Sun, X. Progress in the study of biological effects of hydrogen on higher plants and its promising application in agriculture. Med Gas Res 4, 15 (2014). https://doi.org/10.1186/2045-9912-4-15
https://www.researchgate.net/publication/7114493_Nickel_A_Micronutrient_Essential_for_Higher_Plants
Updated on Sept. 1, 2022
All rights reserved



It was useful
LikeLiked by 1 person